A clinical trial to reduce the treatment time for the Buruli ulcer by half
13 de January de 2022
- Regional Country
- 1.860.033 € Investment
-
2020-25
Proyecto cerradoLength
The Challenge
The Buruli ulcer affects thousands of people in 33 countries and causes disability and stigma.
After decades combatting the Buruli ulcer, which affects thousands of people in over 33 countries, we are still unaware of its exact mode of transmission. We know it is related to environments with stagnant and unsafe water.
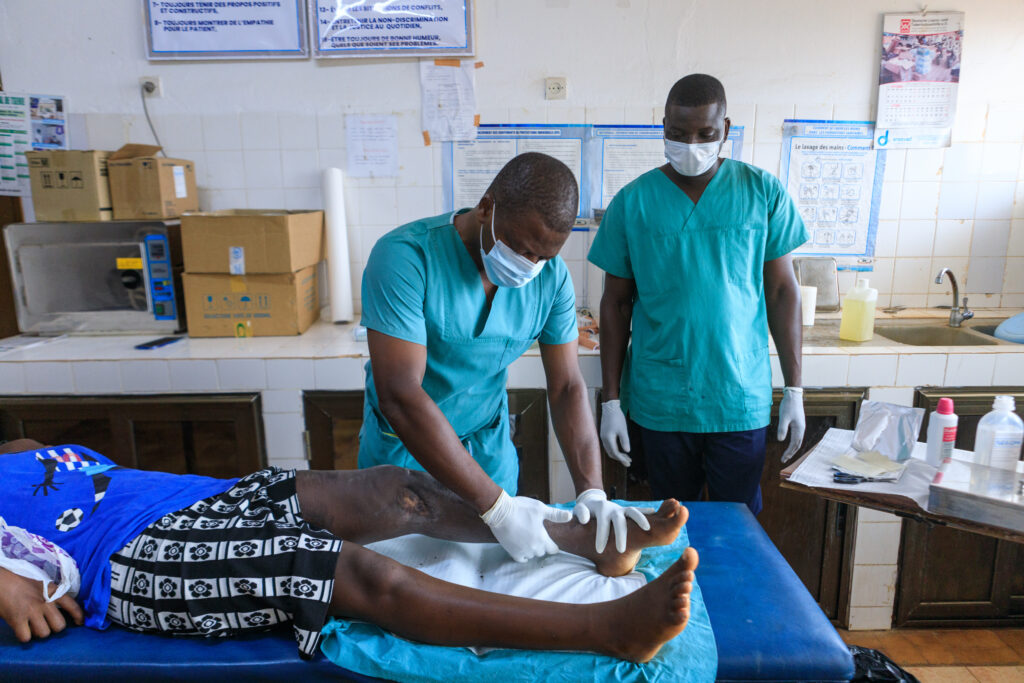
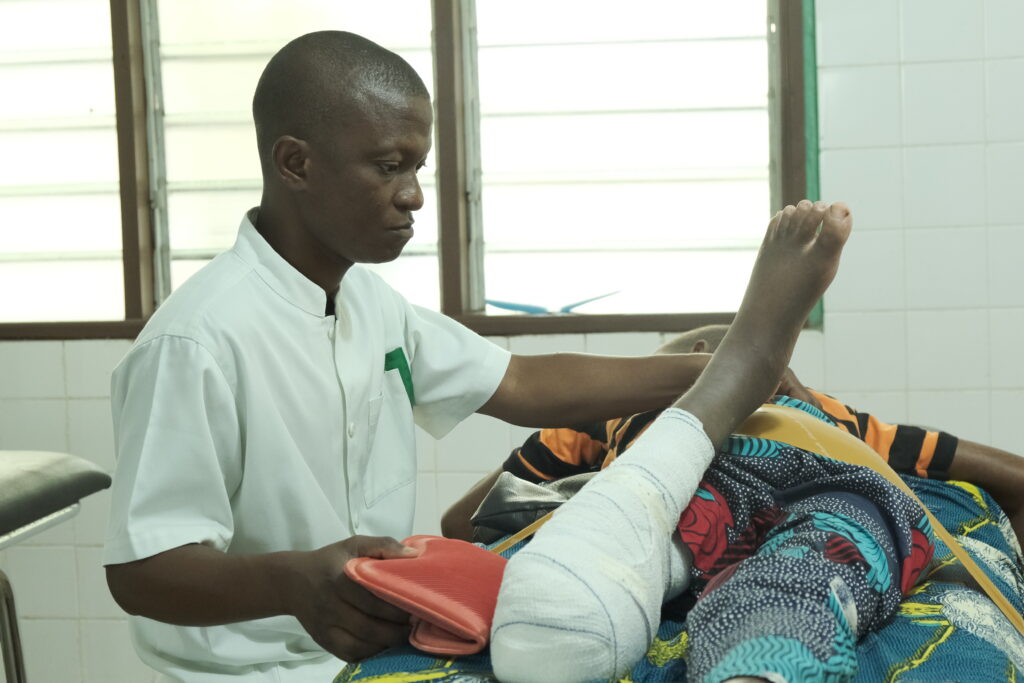
Luckily, the Buruli ulcer has a cure via a treatment combining antibiotics (rifampicin and clarithromycin) over eight weeks. Although in many endemic countries these antibiotics are free, the economic burden of transport, accommodation and food for patients and their carers, and the loss of income and work is too high for those affected and their families, particularly if they require lengthy hospital stays. These expenses affect the health system and families and communities suffering from the disease.
The solution
Through two international research consortiums in Ivory Coast, Ghana, Togo and Benin, we are designing an effective four-week treatment that could reduce healing times and, therefore, expenses related to hospitalisation, travel and care for people suffering from the Buruli ulcer.
- We have implemented a clinical trial in Ivory Coast, Ghana, Togo and Benin.
- We train health staff in implementing procedures of the clinical trial in four countries and the standardisation of diagnosis and wound treatment.
- We strengthen transnational synergies between organisations that work to research and combat NTDs such as the Buruli ulcer.
The consortiums are made up of the Aragón Foundation for Research and Development (ARAID), the University of Zaragoza, the Carlos III Health Institute (ISCIII) and the Anesvad Foundation, from Spain; the Raoul Follereau Foundation (FRF), from France; and the University of Abomey-Calavi (UAC), from Benin, with the support of the Tres Cantos Open Lab Foundation (TCOLF) and GlaxoSmithKline (GSK).
Impact
A positive result in this clinical trial could change the WHO’s guidelines regarding the Buruli ulcer.
A shorter, highly effective and completely oral treatment would reduce the length of the treatment and the healing time of all number of wounds.
Furthermore, this would have an impact on the expenditure related to hospitalisation and the family costs to support the patient.
The insightful information we will obtain could in turn impact strategies to shorten treatments for other infections caused by micro bacteria such as tuberculosis and leprosy.
Related projects
See other projects where we fight Neglected Tropical Diseases